Carbon materials are essential constituents of all lithium-ion (Li-ion) battery systems. In this section we have a closer look at how a Li-ion battery is constructed, the important role of carbon materials in the Li-ion battery formulation, and how keeping these well dispersed can help you realise several processing and performance benefits.

All you need to know about dispersants for carbon in lithium-ion batteries
table of contents
1. How does a Li-ion battery work?
A Li-ion battery is made up of a cathode (positive electrode), an anode (negative electrode), an electrolyte as conductor, and two current collectors (positive and negative). The anode and cathode store the lithium, while the electrolyte carries positively charged lithium ions from the anode to the cathode, and vice versa through the separator. During discharge, the ions flow from the anode to the cathode. Charging reverses the direction, and the ions flow from the cathode to the anode.
The lithium ions' movement creates free electrons in the anode, which creates a charge at the positive current collector. The electrical current then flows from the current collector through the device being powered (cell phone, computer, etc.) to the negative current collector.
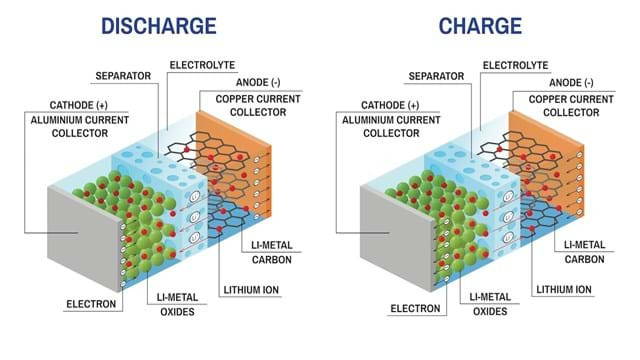
2. What are the main components of the electrodes in a Li-ion battery?
The cathode active material of your Li-ion battery will always consist of a lithium metal oxide, however, the exact chemistry may vary. Typical examples are:
- Lithium Nickel Manganese Cobalt Oxide (LiNiMnCoO2)
- Lithium Cobalt Oxide (LiCoO2)
- Lithium Manganese Oxide (LiMn2O4)
- Lithium Iron Phosphate (LiFePO4)
The choice of active material may impact various battery performance parameters like specific energy, life, thermal stability, and specific power (load capability).
The anode active material of Li-ion batteries is usually based on porous carbon, most commonly graphite. Due to the limited energy density of traditional graphite anodes, alternative anode materials like silicon, sulfur, and even lithium metal are being explored, potentially leading to lithium-ion batteries with 20–40% higher energy density. However, technical challenges related to these anode materials, like swelling of silicon upon charging, have so far, limited their commercial use.
3. What type of carbon materials are used in Li-ion batteries?
As we mentioned above, graphite is the most commonly used anode active material in Li-ion batteries. Graphite is the most stable form of carbon and is the same type of carbon that we find in pencils. Because of its light weight, conductive properties and long-term cycle stability, it is an ideal anode material. Both natural, and synthetic (from petroleum coke) graphite are used for Li-ion anodes, typically in the form of powder or flakes. For optimal battery performance, properties of the graphite are often tailored, including crystallinity, particle size, morphology, and surface chemistry.
Other carbon-based anode materials that may further enhance the performance of Li-ion batteries due to improved electrical, mechanical and thermal properties, but have not yet found commercial use, are graphene and carbon nanotubes (CNTs).
In addition to constituting the anode active material, carbon materials are also used to improve the electrical conductivity of both the positive and negative electrodes, without being involved in the electrochemical redox process, which delivers the energy of the electrochemical cell. These are typically nanosized carbon particles like carbon black and carbon nanofibres, but also include the afore-mentioned graphene, carbon nanotubes and fine graphite powder. Common to all these conductive carbon additives are superior properties of low weight, high chemical inertia and high specific surface area.

Graphite is the most stable form of carbon and is the same type of carbon that we find in lead pencils.
4. Why are conductive carbon materials used in Li-ion battery electrodes?
Conductive carbon additives are essential for optimising the electrical conductivity of battery electrodes. They form a conductive percolation network, filling in the free spaces between the active material particles to increase and maintain the electronic conductivity of the electrode, enabling it to charge and discharge faster. In addition, these conductive additives absorb and retain electrolyte, allowing intimate contact between the lithium ions and active material.
5. How are Li-ion electrodes prepared?
Commercial Li-ion battery electrodes are manufactured by preparing a slurry containing active material, conductive carbon, and a polymer binder in a solvent. The the slurry is then casted onto a metallic current collector (slot coating). The most common solvent is N-Methyl-2-pyrrolidone (NMP), but it can also be water-based.
The binder improves the mechanical stability of the coating, namely the adhesion to the current collector and the mechanical interconnection between adjoining active material particles. The most widely used binder for NMP based slurries is polyvinylidene fluoride (PVDF). Carboxymethyl cellulose (CMC) and Styrene Butadiene Rubber (SBR) are typical binders used for aqueous systems.
When the coating process is complete, the film passes through a dryer to remove the solvent. After drying, the coated foil is cut into narrow strips, which are calendared to reduce thickness variation and increase the film density. Eventually, the cathode electrode film is combined with a separator and the anode film to produce a battery.
6. Why use dispersants for conductive carbon additives?
Conductive carbon additives can have different particle sizes, structures, and porosities. However, all variants have a strong tendency to agglomerate when dispersed in a solvent. To optimise electron flow, and therefore, conductivity, the carbon particles must be uniformly distributed in the electrode slurry prior to casting onto the current collector. You can promote this by adding polymers that help disperse the carbon particles and avoid aggregation.
Dispersants will not only have a positive effect on the conductive carbon additives, but can also aid in dispersing the active graphite material of the electrode. A more uniform distribution of the active material will generally also have a positive effect on battery performance.
In addition to ensuring optimal conductivity, well-dispersed carbon particles reduce the viscosity of your electrode slurry during production, which improves handling and processing. For instance, by avoiding large agglomerated particles, filter clogging can be prevented. Lower viscosity may also allow higher solids loading, which again reduces solvent use.

Dispersants have a positive effect on the conductive carbon additives and help disperse the active graphite material of the electrode.
7. How do better dispersed conductive carbons affect battery performance?
More uniform distribution of carbon particles results in improved electrode conductivity, which translates to improved capacity after many charge and discharge cycles, and longer life of your battery. This is, for instance, important for batteries in electric vehicles (EVs), where potential battery degradation as a result of fast charging may result in the need for consumers to replace their batteries or vehicles prematurely.
8. What are typical dispersants for carbon materials in Li-ion electrodes?
In many cases, the polymer binder in the electrode slurry acts also as a dispersant for the conductive carbon particles in the formulation. The previously mentioned polyvinylidene fluoride (PVDF), carboxymethyl cellulose (CMC), and Styrene Butadiene Rubber (SBR) will all to some degree contribute to a more even distribution of conductive carbons.
However, since these products are primarily tailored to hold the active material particles together and in contact with the current collectors, they may not provide optimal dispersing effect.
Therefore, it is often beneficial to add a dispersing aid specifically designed to prevent agglomeration and ensure uniform distribution of carbon particles during slurry preparation. Examples of such dispersants include:
- block co-polymers
- naphthalene sulfonates
- lignosulfonates





